Voluntary withdrawals of GAMUNEX-C 10% lots
Prescribe GAMUNEX-C to deliver the power of proven neuroprotection from inflammation in CIDP1,2
The longest randomized study of IVIG in CIDP2,3
The ICE study demonstrated that GAMUNEX-C delivered early and sustained improvements in CIDP symptoms with >87% of responders remaining relapse-free at 48 weeks
#1 prescribed IG for CIDP1,4,5
An IVIG disrupts inflammation through multiple pathways
The precise mechanism of action of GAMUNEX-C in the treatment of CIDP has not been fully elucidated
Maximum purity1,6-9
The unique manufacturing process yields the maximum percentage of IgG (≥98%) while minimizing aggregates that may lead to tolerability concerns
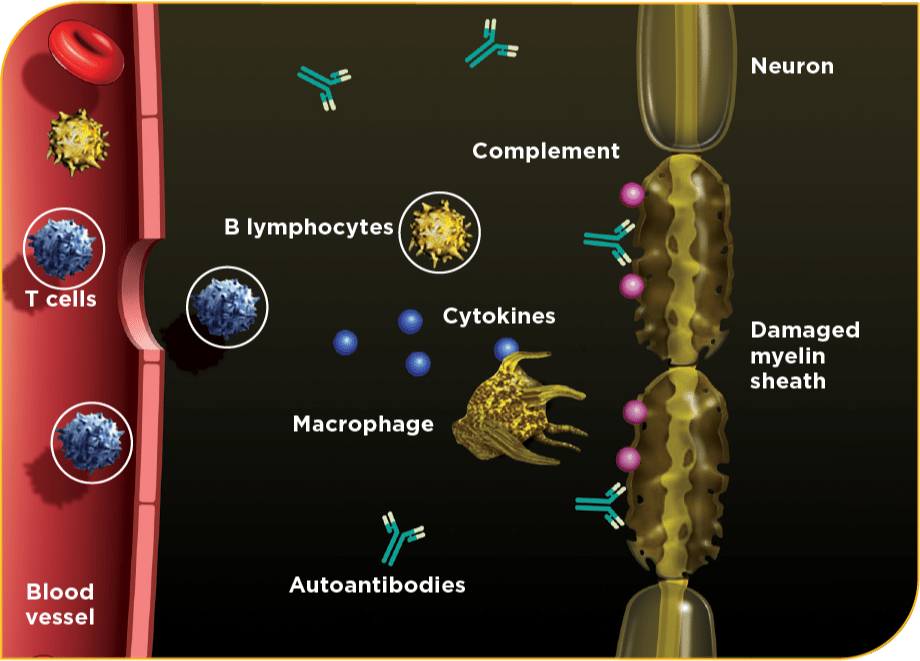
CIDP is a complex inflammatory condition with various suspected pathogenic mechanisms10
Peer-to-Peer Discussions
Join us for our upcoming live and virtual events.
Gamunex Connexions
Take advantage of resources for your office and find support and financial assistance for your patients.
Clinical Resources
Access publications, resources, and links to organizations for the latest insights.
Receive updates and important information on GAMUNEX-C
Important Safety Information
GAMUNEX®-C (immune globulin injection [human], 10% caprylate/chromatography purified) is indicated for the treatment of primary humoral immunodeficiency disease (PIDD) in patients 2 years of age and older, idiopathic thrombocytopenic purpura (ITP) in adults and children, and chronic inflammatory demyelinating polyneuropathy (CIDP) in adults.
Thrombosis may occur with immune globulin products, including GAMUNEX-C. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling central vascular catheters, hyperviscosity, and cardiovascular risk factors. Thrombosis may occur in the absence of known risk factors. For patients at risk of thrombosis, administer GAMUNEX-C at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity.
Renal dysfunction, acute renal failure, osmotic nephrosis, and death may occur with immune globulin intravenous (IVIG) products in predisposed patients. Patients predisposed to renal dysfunction include those with any degree of preexisting renal insufficiency, diabetes mellitus, age greater than 65, volume depletion, sepsis, paraproteinemia, or patients receiving known nephrotoxic drugs. Renal dysfunction and acute renal failure occur more commonly in patients receiving IVIG products containing sucrose. GAMUNEX-C does not contain sucrose. For patients at risk of renal dysfunction or failure, administer GAMUNEX-C at the minimum concentration available and the minimum infusion rate practicable.
GAMUNEX-C is contraindicated in patients who have had an anaphylactic or severe systemic reaction to the administration of human immune globulin. It is contraindicated in IgA-deficient patients with antibodies against IgA and history of hypersensitivity.
Severe hypersensitivity reactions may occur with IVIG products, including GAMUNEX-C. In case of hypersensitivity, discontinue GAMUNEX-C infusion immediately and institute appropriate treatment.
Monitor renal function, including blood urea nitrogen (BUN), serum creatinine, and urine output in patients at risk of developing acute renal failure.
Hyperproteinemia, increased serum viscosity, and hyponatremia may occur in patients receiving IVIG treatment, including GAMUNEX-C.
There have been reports of aseptic meningitis, hemolytic anemia, and noncardiogenic pulmonary edema (transfusion-related acute lung injury [TRALI]) in patients administered with IVIG, including GAMUNEX-C.
The high-dose regimen (1g/kg x 1-2 days) is not recommended for individuals with expanded fluid volumes or where fluid volume may be a concern.
Because GAMUNEX-C is made from human blood, it may carry a risk of transmitting infectious agents, eg, viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent.
Do not administer GAMUNEX-C subcutaneously in patients with ITP because of the risk of hematoma formation.
Periodic monitoring of renal function and urine output is particularly important in patients judged to be at increased risk of developing acute renal failure. Assess renal function, including measurement of BUN and serum creatinine, before the initial infusion of GAMUNEX-C and at appropriate intervals thereafter.
Consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia/markedly high triacylglycerols (triglycerides), or monoclonal gammopathies, because of the potentially increased risk of thrombosis.
If signs and/or symptoms of hemolysis are present after an infusion of GAMUNEX-C, perform appropriate laboratory testing for confirmation.
If TRALI is suspected, perform appropriate tests for the presence of antineutrophil antibodies and anti-HLA antibodies in both the product and patient's serum.
After infusion of IgG, the transitory rise of the various passively transferred antibodies in the patient's blood may yield positive serological testing results, with the potential for misleading interpretation.
In clinical studies, the most common adverse reactions with GAMUNEX-C were headache, pyrexia, hypertension, chills, rash, nausea, arthralgia, and asthenia (in CIDP); cough, rhinitis, pharyngitis, headache, asthma, nausea, fever, diarrhea, and sinusitis with intravenous use (in PIDD) and local infusion-site reactions, fatigue, headache, upper respiratory tract infection, arthralgia, diarrhea, nausea, sinusitis, bronchitis, depression, allergic dermatitis, migraine, myalgia, viral infection, and pyrexia with subcutaneous use (in PIDD); and headache, ecchymosis, vomiting, fever, nausea, rash, abdominal pain, back pain, and dyspepsia (in ITP).
The most serious adverse reactions in clinical studies were pulmonary embolism (PE) in 1 subject with a history of PE (in CIDP), an exacerbation of autoimmune pure red cell aplasia in 1 subject (in PIDD), and myocarditis in 1 subject that occurred 50 days post-study drug infusion and was not considered drug related (in ITP).
Please see accompanying full Prescribing Information for GAMUNEX-C.
Terms to know
CIDP, chronic inflammatory demyelinating polyneuropathy; IG, immune globulin; IgG, immunoglobulin G; IVIG, intravenous immunoglobulin.
References
- GAMUNEX®-C (immune globulin injection [human], 10% caprylate/chromatography purified) Prescribing Information. Grifols.
- Hughes RAC, Donofrio P, Bril V, et al; on behalf of the ICE Study Group. Intravenous immune globulin (10% caprylate chromatography purified) for the treatment of chronic inflammatory demyelinating polyradiculoneuropathy (ICE study): a randomized placebo-controlled trial. Lancet Neurol. 2008;7(2):136-144.
- Donofrio PD, Bril V, Dalakas MC, et al. Safety and tolerability of immune globulin intravenous in chronic inflammatory demyelinating polyradiculoneuropathy [published correction appears in Arch Neurol. 2010 Dec; 67(12):1515]. Arch Neurol. 2010;67(9):1082-1088.
- Data on file, Grifols.
- Durandy A, Kaveri SV, Kuijpers TW, et al. Intravenous immunoglobulins – understanding properties and mechanisms. Clin Exp Immunol. 2009;158(suppl1):2-13.
- Lebing W, Remington KM, Schreiner C, Paul HI. Properties of a new intravenous immunoglobulin (IGIV-C, 10%) produced by virus inactivation with caprylate and column chromatography. Vox Sang. 2003;84(3):193-201.
- Alonso W, Vandeberg P, Lang J, et al. Immune globulin subcutaneous, human 20% solution. Biologicals. 2020;64:34-40.
- Schwab I, Nimmerjahn F. Intravenous immunoglobulin therapy: how does IgG modulate the immune system? Nat Rev Immunol. 2013;13(3):176-189.
- Bertolini J. The purification of plasma proteins for therapeutic use. In: Simon TL, ed. Rossi’s Principles of Transfusion Medicine. 5th ed. John Wiley & Sons; 2016:302-320.
- Khoo A, Frasca J, Schultz D. Measuring disease activity and predicting response to intravenous immunoglobulin in chronic inflammatory demyelinating polyneuropathy. Biomark Res. 2019;7:3.