
Learn about a 20% subcutaneous IG option for your PIDD patients
XEMBIFY® is a 20% immune globulin indicated for treatment of primary humoral immunodeficiency disease (PIDD) in patients 2 years of age and older. XEMBIFY is for subcutaneous administration only.
Coverage authorization guidance and support
Utilize these guides to help you and your staff with your patients' coverage authorization, appeals, and copay assistance.
GAMUNEX-C Coverage Authorization Letters
A guide for your practice on the coverage authorization process, including a sample Authorization Appeals Letter, Letter of Medical Necessity, and Formulary Exception Request.
An overview on how to submit claims for your eligible patients through the GAMUNEX Copay Assistance Program.
Coding and reimbursement guidance
Health insurance reimbursement policies can be complex and confusing. Gamunex Connexions® can help you by providing easy-to-understand information on coverage, coding, reimbursement, and claims submissions.
For technical support regarding the Copay Assistance Program, call 1-866-ADHERE2 (1-866-234-3732) (available 9 AM to 8 PM ET) or email support@medmonk.com.
If you have any other questions regarding GAMUNEX-C or clinical information, contact a Grifols Sales Representative.
GAMUNEX-C packaging and storage
Convenient packaging and storage offered by GAMUNEX-C1
- Built-in hanger on 5, 10, 20, and 40 g vials
- Long 3-year shelf life at 2-8°C (36-46°F) from the date of manufacture
- Room temperature storage (not to exceed 25°C or 77°F) for up to 6 months at any time during 36-month shelf life
- Latex-free
- 6 color-coded package sizes with barcode
- Vials available in 1, 2.5, 5, 10, 20, and 40 g
- Peel-off labels to simplify record keeping
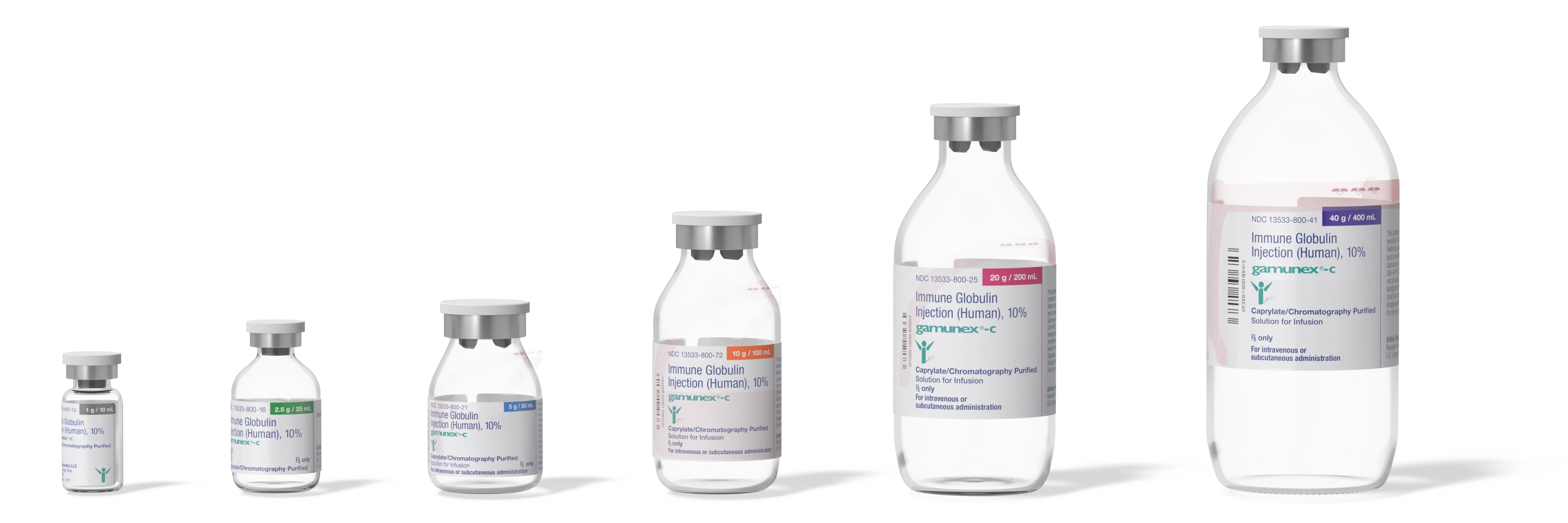
Not actual size.
Order it from GAMUNEX-C Customer Service:
1-800-243-4153
Get it shipped to:
Home healthcare/specialty pharmacies
Specialty biological distributors
Directly to the physician or patient
Important Safety Information
XEMBIFY® (immune globulin subcutaneous, human–klhw), 20% and GAMUNEX®-C (immune globulin injection [human], 10% caprylate/chromatography purified) are immune globulins indicated for treatment of primary humoral immunodeficiency disease (PIDD) in patients 2 years of age and older.
XEMBIFY is a 20% solution for subcutaneous administration only. GAMUNEX-C is a 10% solution for intravenous and subcutaneous administration in PIDD.
Important Safety Information
WARNING: THROMBOSIS
Thrombosis may occur with immune globulin products, including XEMBIFY and GAMUNEX-C. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, estrogens, indwelling vascular catheters, hyperviscosity, and cardiovascular risk factors. Thrombosis may occur in the absence of known risk factors.
For patients at risk of thrombosis, administer XEMBIFY and GAMUNEX-C at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk of hyperviscosity.
WARNING: RENAL DYSFUNCTION and ACUTE RENAL FAILURE
Renal dysfunction, acute renal failure, osmotic nephrosis, and death may occur with GAMUNEX-C in predisposed patients. Patients predisposed to renal dysfunction include those with any degree of preexisting renal insufficiency, diabetes mellitus, age greater than 65, volume depletion, sepsis, paraproteinemia, or patients receiving known nephrotoxic drugs.
Renal dysfunction and acute renal failure occur more commonly in patients receiving IVIG products containing sucrose. GAMUNEX-C does not contain sucrose.
For patients at risk of renal dysfunction or failure, administer GAMUNEX-C at the minimum concentration available and the minimum infusion rate practicable.
Contraindications
XEMBIFY and GAMUNEX-C are contraindicated in:
Patients who have had an anaphylactic or severe systemic reaction to the administration of human immune globulin.
IgA-deficient patients with antibodies against IgA and a history of hypersensitivity.
Warnings and Precautions
Hypersensitivity. Severe hypersensitivity reactions may occur with immune globulin products, including XEMBIFY and GAMUNEX-C. In case of hypersensitivity, discontinue infusion immediately and institute appropriate treatment. XEMBIFY and GAMUNEX-C contain IgA. Patients with known antibodies to IgA may have a greater risk of developing potentially severe hypersensitivity and anaphylactic reactions.
Aseptic meningitis syndrome (AMS). AMS may occur with human immune globulin treatment, including XEMBIFY and GAMUNEX-C. Conduct a thorough neurological exam on patients exhibiting signs and symptoms of AMS to rule out other causes of meningitis. Discontinuation of treatment has resulted in remission within several days without sequelae.
Thrombosis. Thrombosis may occur following treatment with immune globulin products, including XEMBIFY and GAMUNEX-C, even in the absence of known risk factors. In patients at risk, administer at the minimum dose and infusion rate practicable. Ensure adequate hydration before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk of hyperviscosity.
Hyperproteinemia, Increased Serum Viscosity, and Hyponatremia. Hyperproteinemia, increased serum viscosity, and hyponatremia may occur in patients receiving IVIG treatment, including GAMUNEX-C.
Renal dysfunction/failure. Acute renal dysfunction/failure, acute tubular necrosis, proximal tubular nephropathy, osmotic nephrosis, and death may occur with use of human immune globulin products, especially those containing sucrose. XEMBIFY and GAMUNEX-C do not contain sucrose. Ensure patients are not volume-depleted prior to starting infusion. In patients at risk due to preexisting renal insufficiency or predisposition to acute renal failure, assess renal function (including blood urea nitrogen [BUN], serum creatinine, and urine output) prior to the initial infusion and again at appropriate intervals thereafter. If renal function deteriorates, consider discontinuation.
Hemolysis. XEMBIFY and GAMUNEX-C may contain blood group antibodies that may cause a positive direct antiglobulin reaction and hemolysis. Monitor patients for clinical signs and symptoms of hemolysis. If signs and symptoms are present after infusion, perform confirmatory lab testing.
Transfusion-related acute lung injury (TRALI). Noncardiogenic pulmonary edema may occur in patients following treatment with immune globulin products, including XEMBIFY and GAMUNEX-C. Monitor patients for pulmonary adverse reactions. If TRALI is suspected, perform appropriate tests for the presence of antineutrophil and anti-HLA antibodies in both the product and patient serum. TRALI may be managed using oxygen therapy with adequate ventilatory support.
Transmissible infectious agents. Because XEMBIFY and GAMUNEX-C are made from human blood, they may carry a risk of transmitting infectious agents, eg, viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent.
Interference with lab tests. Passively transferred antibodies in the patient’s blood may yield positive serological testing results, with the potential for misleading interpretation.
Adverse Reactions
The most common adverse reactions in ≥ 5% of subjects in clinical trials were:
XEMBIFY
PIDD, subcutaneous: local adverse reactions, including infusion-site erythema (redness), infusion-site pain, infusion-site swelling (puffiness), infusion-site bruising, infusion-site nodule, infusion-site pruritus (itching), infusion-site induration (firmness), infusion-site scab, infusion-site edema, and systemic reactions including cough and diarrhea.
GAMUNEX-C
PIDD, subcutaneous: local infusion-site reactions, fatigue, headache, upper respiratory tract infection, arthralgia, diarrhea, nausea, sinusitis, bronchitis, depression, allergic dermatitis, migraine, myalgia, viral infection, and pyrexia.
PIDD, intravenous: cough, rhinitis, pharyngitis, headache, asthma, nausea, fever, diarrhea, and sinusitis.
The most serious adverse reaction in clinical studies with GAMUNEX-C in PIDD was an exacerbation of autoimmune pure red cell aplasia in 1 subject.
Drug Interactions
Passive transfer of antibodies may transiently interfere with the immune responses to live attenuated virus vaccines (eg, measles, mumps, rubella, and varicella).
Please see accompanying full Prescribing Information for XEMBIFY.
Please see accompanying full Prescribing Information for GAMUNEX-C.
Terms to know
CIDP, chronic inflammatory demyelinating polyneuropathy; ICE, caprylate-chromatography purified immune globulin intravenous (IGIV-C) CIDP efficacy; IG, immune globulin; ITP, idiopathic thrombocytopenic purpura; IV, intravenous; PIDD, primary immunodeficiency disease; Sub Q, subcutaneous.
Reference
- GAMUNEX®-C (immune globulin injection [human], 10% caprylate/chromatography purified) Prescribing Information. Grifols.